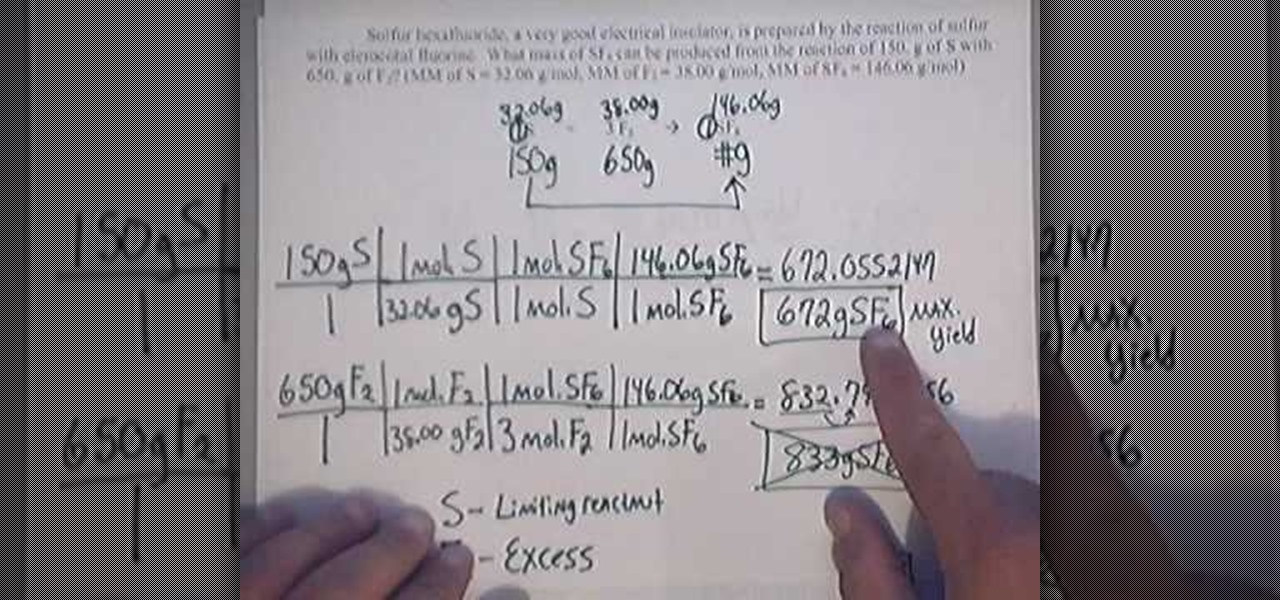Exemplary Info About How To Improve Percent Yield

Reading through an analytical textbook, it states a few ways to improve yield and purity but it doesn't actually state how you would do it practically:
How to improve percent yield. To get percent yield, insert the values in the percent yield formula, so the percent yield equation will be. For example, consider the decomposition process of calcium carbonate. Deduct the property's ongoing costs and costs of vacancy (i.e lost.
To increase the percentage yield of ethyl ethanoate can be achieved by removing water or ester during esterification. Since the only inputs in the formula to calculate percentage yield are the return and the investment cost, the only way to increase the percentage yield is to either. At higher temperatures, the kinetic energy of the reactant molecules will increase, and this would lead to.
How to work with thiols. P = (a / t) × 100 %. Tips for flash column chromatography.
Veratrole was the limiting reagent and it was. I have seen many grignard reactions performed on the large scale…and in the. * remove 1 or more products as the reaction.
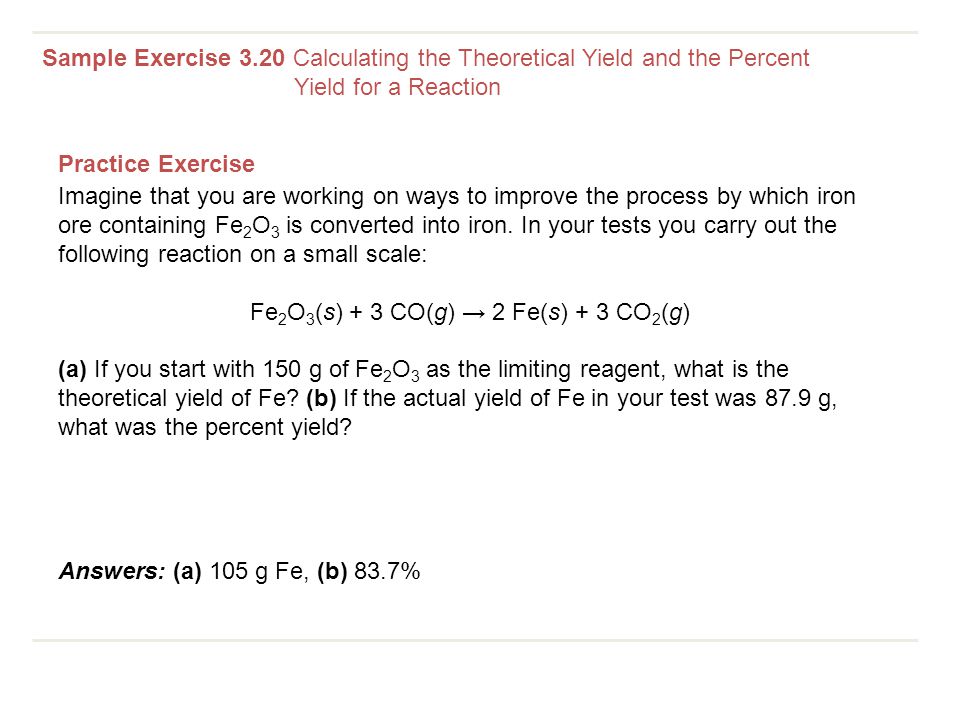
And with what substrate do you react it? * mix different amounts of the two chemicals. Percent yield = theoretical yield ÷ actual yield × 100.
At higher temperatures, the kinetic energy of the reactant molecules will increase, and this. Restaurant inventory management software can help you track food waste and yield. Where, p is the percentage yield, a is the actual yield, t is the theoretical yield.
Simple things to try : Well, what grignard are you making? * change reaction conditions (temprature or pressure) * use a catalyst.
To calculate yield, you need to follow a few steps to get the property's yield as an annual percentage. Percent yield = theoretical yield ÷ actual yield × 100. Allow the solution cool down as slowly as.
Huge gains in yield have been achieved through lean manufacturing, six sigma and kaizen — so much that it may seem no more can be done, particularly in the waste. How to increase the percentage yield of a reaction? Alternatively, one of your reagents was limiting, and caused a reduced yield.
How to increase the percentage yield of a reaction?